
Get Instant Solution By an Expert Advisor
(4.8)


India’s medical device and drug regulatory framework is governed by the Central Drugs Standard Control Organization, which operates under the Ministry of Health and Family Welfare. With the growing importance of medical devices and pharmaceuticals, CDSCO has established a structured system of forms and licenses to regulate manufacturing, import, testing, and approval processes.
Among these, Form MD 3, MD 5, MD 7, and MD 9 are commonly used in regulatory submissions related to medical devices. Understanding these forms is essential for manufacturers, importers, and regulatory professionals.
CDSCO forms are standardized applications and approvals used under the Medical Device Rules, 2017. These forms streamline processes such as obtaining licenses, approvals, and permissions for different stages of the medical device lifecycle.
Each form serves a specific purpose—ranging from import licensing to manufacturing approvals and testing permissions.
MD 42 License Registration in Delhi NCR and Haryana
Form MD 3 is used to apply for an import license for medical devices in India.
If a company wants to import medical devices for sale, distribution, or use in India, Form MD 3 must be submitted.
This form is crucial because it ensures that imported devices meet Indian safety and quality standards.
Form MD 5 is used for obtaining a manufacturing license for medical devices in India.
Any company intending to manufacture medical devices in India must apply through this form.
Form MD 5 plays a critical role in ensuring that locally manufactured devices comply with regulatory norms and quality standards.
Form MD 7 is used to apply for a loan license.
A loan license allows a company to manufacture medical devices using another manufacturer’s facilities.
This form is particularly useful for startups or companies looking to enter the market without heavy infrastructure investment.
Form MD 9 is used for obtaining a test license.
Before launching a product, companies often need to test devices for safety and performance. Form MD 9 enables this legally under CDSCO regulations.
MD 15: CDSCO Medical Device Import License?
| Form | Purpose | Applicant Type | Usage Stage |
|---|---|---|---|
| MD 3 | Import License | Importers | Market Entry |
| MD 5 | Manufacturing License | Manufacturers | Production |
| MD 7 | Loan License | Third-party companies | Outsourced Production |
| MD 9 | Test License | R&D / Importers | Testing & Evaluation |
The general application process for these forms includes:
Understanding Form MD 3, MD 5, MD 7, and MD 9 is essential for navigating India’s medical device regulatory landscape. Each form serves a unique purpose—from importing and manufacturing to testing and outsourcing production.
Whether you are a manufacturer, importer, or startup entering the healthcare sector, compliance with CDSCO regulations ensures not only legal approval but also the safety and reliability of medical devices in the Indian market.
If approached correctly, these forms become less of a regulatory burden and more of a structured pathway to market success.

 Nishi Chawla
Nishi Chawla
22 Apr, 2026

 Nishi Chawala
Nishi Chawala
22 Apr, 2026

 Nishi Chawla
Nishi Chawla
21 Apr, 2026
(2).webp)
 Nishi Chawla
Nishi Chawla
21 Apr, 2026
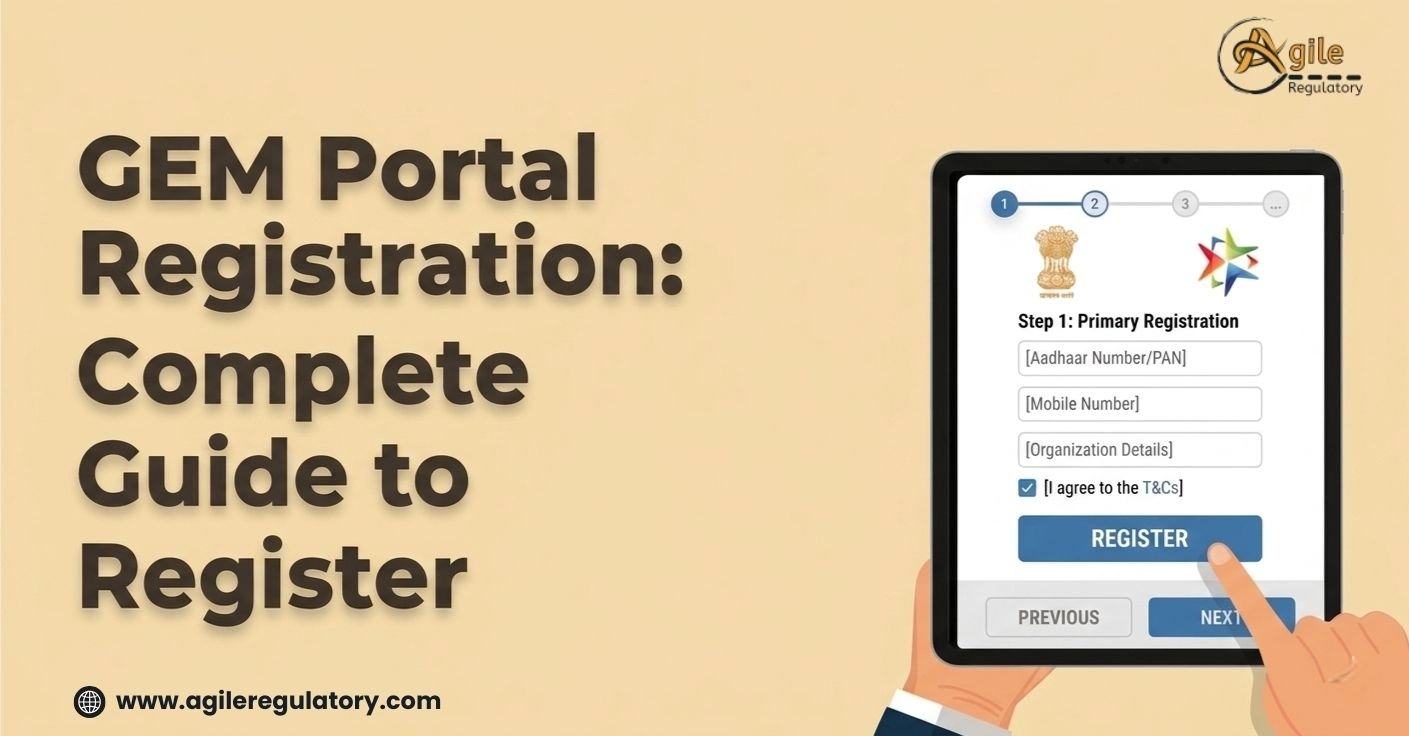
 Nishi Chawla
Nishi Chawla
20 Apr, 2026

Get Instant Solution By an Expert Advisor
(4.8)
We simplify compliance through a proven 4-step process: Consultation, Documentation, Submission, and certification. From understanding requirements to getting final approvals, we deliver a smooth, timely, and fully compliant journey for your business.
What our customer says about us
Fantastic support from the team. Their expertise transformed our approach, driving remarkable outcomes. A must-have partner for businesses seeking effective consulting solutions. Highly recommended.

KTPL Instruments
Agile Regualtory delivers exceptional solutions. Their insightful guidance streamlined our processes and boosted profitability. Highly recommended for businesses seeking expert consulting services to thrive.

Justrack IOT
Impressed by Agile Regulatory's expertise. Their strategic insights and practical solutions have elevated our business operations. A reliable partner for effective consulting services. Highly recommended for growth-focused businesses.

Coaire Compressor
Extraordinary consulting services. Their insightful solutions and dedicated team reshaped our business, driving remarkable improvements. Highly recommend it for transformative results.

Easy Polymer
Incredible experience with Agile Regulatory. Their innovative strategies and expert advice revitalized our business model, resulting in impressive growth. Highly recommend their exceptional consulting services.

Tarus International
Top-tier consulting! offered strategic solutions that revolutionized our approach. Their deep expertise and personalized guidance made a significant impact on our success. Highly recommend their services.

Anchor Weighing
Agile Regulatory exceeded expectations! Their tailored solutions, expertise, and proactive approach led to remarkable results. Highly recommend for businesses seeking impactful and strategic guidance.

AM Capacitor
Outstanding service! delivered targeted solutions with professionalism and expertise. Their insights elevated our business strategies, resulting in noticeable growth. Highly recommended for exceptional consultation.
Imaxx Pro Aquistic
Leave a Reply
Your email address will not be published. Required fields are marked *